MedTech Startup’s 12-Month Clinical Trial Roadmap 2026

MedTech startups can successfully transition from concept to clinical trial within 12 months in 2026 by meticulously planning regulatory strategies, securing funding, developing robust prototypes, and engaging key stakeholders early in the process.
Embarking on the journey to bring a groundbreaking medical device to market is an ambitious undertaking, particularly for emerging companies. For MedTech startups, navigating the complex landscape from initial idea to a successful clinical trial within a tight timeframe requires precise planning and execution. This article presents a detailed From Concept to Clinical Trial: A 12-Month Roadmap for MedTech Startups in 2026, offering a strategic guide for founders aiming to accelerate their innovations while ensuring regulatory compliance and patient safety.
Laying the Foundation: Months 1-2 – Ideation, Research, and Initial Design
The initial phase of any MedTech venture is critical, setting the stage for all subsequent developments. Months one and two are dedicated to solidifying the core concept, conducting thorough research, and initiating the preliminary design of the medical device. This period requires a deep dive into unmet medical needs, existing solutions, and potential market opportunities.
Understanding the problem your device aims to solve, and for whom, is paramount. This involves extensive literature reviews, consultations with medical professionals, and patient interviews to gather qualitative and quantitative data. The goal is to define the device’s intended use, target user, and preliminary specifications. This early clarity helps prevent costly redesigns later on.
Market Analysis and Competitive Landscape
Before committing significant resources, a comprehensive market analysis is essential. This includes identifying your target market, understanding its size and growth potential, and assessing the competitive landscape. Knowledge of direct and indirect competitors allows you to differentiate your product effectively.
- Identify key competitors and their product offerings.
- Analyze market trends and regulatory shifts impacting your niche.
- Determine the unique selling proposition (USP) of your device.
- Assess potential barriers to market entry.
Simultaneously, intellectual property (IP) considerations must be addressed. A preliminary patent search helps identify existing patents that could impact your device and informs your IP strategy. Protecting your innovation from the outset is crucial for long-term success and investor confidence. This foundational work ensures that the concept is not only innovative but also viable and defensible in the marketplace.
By the end of this phase, a clear concept document, initial design specifications, and a robust understanding of the market and competitive environment should be in place, providing a solid launchpad for the next steps.
Regulatory Strategy and Funding: Months 3-4 – Navigating the US FDA Landscape
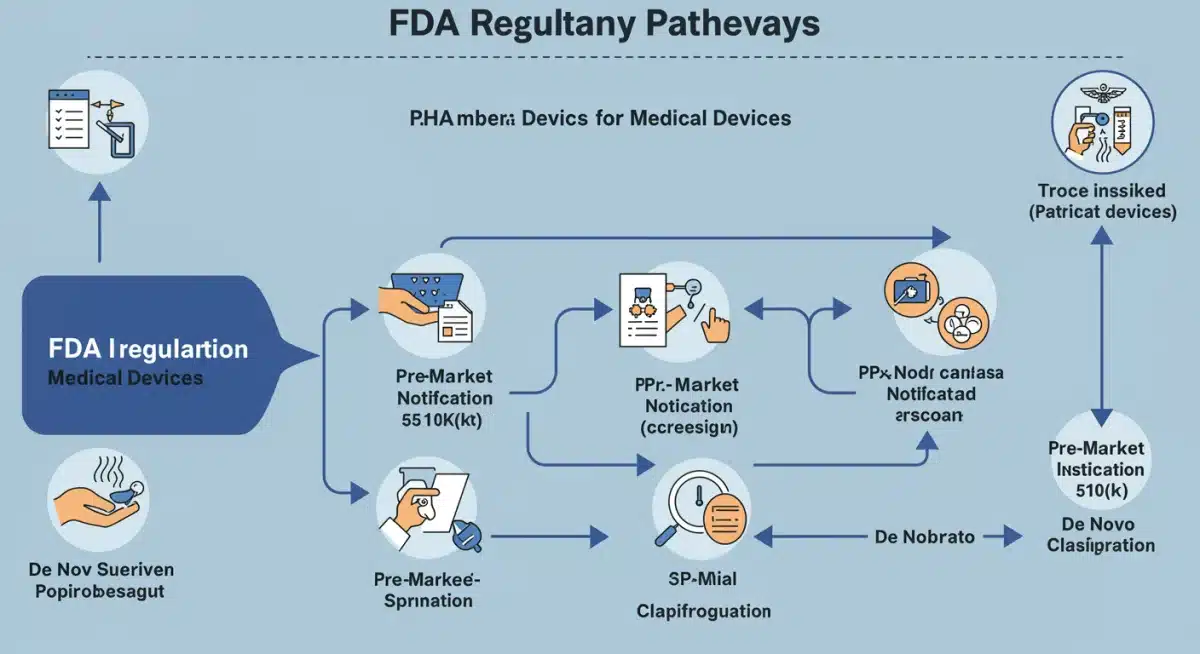
Months three and four are focused on establishing a clear regulatory pathway and securing the necessary financial resources. These two elements are intrinsically linked, as investors often require a well-defined regulatory strategy before committing capital. The US Food and Drug Administration (FDA) is the primary regulatory body, and understanding its requirements is non-negotiable for MedTech startups.
Developing an early regulatory strategy involves classifying your device, identifying the appropriate regulatory pathway (e.g., 510(k), PMA, De Novo), and understanding the specific data requirements for submission. Engaging regulatory consultants at this stage can be invaluable, especially for nascent teams without extensive regulatory experience. Their expertise can help streamline the process and avoid common pitfalls that delay market entry.
Securing Seed Funding and Grant Opportunities
Funding is the lifeblood of any startup, and for MedTech, the capital requirements are often substantial due to research, development, and clinical trial costs. During this period, startups should actively pursue seed funding rounds, angel investors, and non-dilutive grants.
- Prepare a compelling business plan and investor deck.
- Identify and network with relevant angel investors and venture capitalists specializing in MedTech.
- Research and apply for Small Business Innovation Research (SBIR) or Small Business Technology Transfer (STTR) grants.
- Develop a detailed financial projection, including R&D, regulatory, and clinical trial costs.
A well-articulated value proposition, a clear regulatory plan, and a strong team are key factors that attract investors. Demonstrating a deep understanding of the market opportunity and a credible path to commercialization will significantly enhance fundraising efforts. This dual focus on regulatory strategy and funding ensures the startup is both compliant and financially equipped for the demanding development process ahead.
By the close of this period, the startup should have a foundational regulatory strategy, potentially with initial FDA consultations underway, and a clear financial runway to support the next phases of development.
Prototype Development and Verification: Months 5-6 – Bringing the Concept to Life
With a solid concept, regulatory strategy, and initial funding in place, months five and six are dedicated to the tangible development of the device. This involves creating functional prototypes and rigorously testing them to ensure they meet design specifications and performance requirements. This phase is iterative, often involving multiple rounds of design, build, and test cycles.
The engineering team will translate the initial design specifications into a working prototype. This includes selecting appropriate materials, developing manufacturing processes, and integrating software components if applicable. Emphasis should be placed on creating a device that is not only functional but also manufacturable and scalable.
Design Control and Risk Management
Adherence to design control principles is crucial during prototype development. This involves documenting all design inputs, outputs, reviews, verifications, and validations. A robust quality management system (QMS) should be implemented, even at this early stage, to ensure compliance with ISO 13485 standards.
- Implement a comprehensive design control process.
- Conduct thorough risk assessments (e.g., FMEA) to identify and mitigate potential hazards.
- Perform verification testing to confirm design outputs meet design inputs.
- Document all testing procedures and results meticulously.
Early verification testing helps identify design flaws and areas for improvement before significant resources are committed to later stages of development. This proactive approach minimizes the chances of encountering major issues during the more expensive validation and clinical trial phases. The goal is to produce a refined prototype that is ready for formal validation and pre-clinical studies.
At the end of this two-month period, the MedTech startup should possess a functional and verified prototype, accompanied by comprehensive design documentation and a robust risk management file, setting the stage for pre-clinical evaluations.
Pre-clinical Testing and Quality System Implementation: Months 7-8 – Ensuring Safety and Efficacy
Months seven and eight involve critical pre-clinical testing and the full implementation of a robust quality management system (QMS). Pre-clinical studies are essential to gather data on the device’s safety and preliminary efficacy before it can be tested in humans. These studies often include bench testing, in vitro studies, and animal studies, depending on the device’s classification and intended use.
Selecting the right contract research organizations (CROs) for pre-clinical testing is paramount. These partners must have proven expertise and operate under Good Laboratory Practice (GLP) regulations. The data generated from these studies will form a crucial part of your regulatory submission to the FDA.
Establishing a Comprehensive Quality Management System
A fully operational QMS is not just a regulatory requirement; it’s a cornerstone of responsible device development. This period should see the QMS moving beyond initial implementation to active use across all aspects of the startup’s operations.
- Finalize QMS documentation, including SOPs, work instructions, and quality records.
- Conduct internal audits to ensure QMS effectiveness and compliance.
- Train all relevant personnel on QMS procedures and their roles.
- Prepare for external QMS audits, such as ISO 13485 certification, if applicable.
The data from pre-clinical testing will be meticulously analyzed and summarized in a comprehensive report. This report, along with the established QMS, demonstrates the device’s safety profile and the startup’s commitment to quality. Successfully completing this phase means the device is scientifically and systematically prepared for human trials, a major milestone in the development roadmap.
By the end of month eight, the startup should have compelling pre-clinical data supporting the device’s safety and preliminary performance, coupled with a fully implemented and actively utilized QMS, ready for regulatory scrutiny.
Regulatory Submission and Clinical Protocol Development: Months 9-10 – Seeking FDA Approval for Trials
Months nine and ten are pivotal, focusing on compiling the regulatory submission and meticulously developing the clinical trial protocol. This is where all the prior hard work converges into a formal application to the FDA, typically an Investigational Device Exemption (IDE) application for significant risk devices, or a Non-Significant Risk (NSR) determination for lower-risk devices.
The IDE submission requires a comprehensive package including pre-clinical data, manufacturing information, risk analysis, and the proposed clinical protocol. Accuracy and completeness are critical; any deficiencies can lead to delays or outright rejection. This phase demands strong project management to ensure all documents are properly compiled and cross-referenced.
Crafting a Robust Clinical Trial Protocol
Simultaneously, the clinical trial protocol must be developed in detail. This document outlines every aspect of how the clinical study will be conducted, ensuring scientific rigor and ethical compliance.
- Define study objectives, endpoints, and statistical considerations.
- Establish clear inclusion and exclusion criteria for study participants.
- Detail the study design (e.g., randomized, controlled, single-arm).
- Outline data collection methods, adverse event reporting, and data management plans.
The protocol must also address ethical considerations, including informed consent processes and patient confidentiality, aligning with Good Clinical Practice (GCP) guidelines. Collaboration with clinical experts, biostatisticians, and ethicists is essential to develop a protocol that is scientifically sound and ethically defensible. A well-designed protocol is key to generating reliable data that supports the device’s safety and efficacy claims.
At the close of this period, the MedTech startup should have submitted its regulatory application to the FDA and finalized a comprehensive clinical trial protocol, marking the crucial transition from pre-clinical development to the cusp of human studies.
Site Selection, IRB Approval, and Trial Launch: Months 11-12 – The Final Push to Clinical Trials
The final two months of the roadmap are dedicated to securing Institutional Review Board (IRB) approval, selecting clinical trial sites, and ultimately launching the clinical trial. This phase is characterized by intense coordination and communication among various stakeholders.
Upon receiving FDA approval for the IDE (or NSR determination), the focus shifts to obtaining IRB approval for each participating clinical site. The IRB reviews the clinical protocol, informed consent forms, and other study-related documents to ensure the protection of human subjects’ rights and welfare. This process can be iterative, requiring responses to IRB questions and modifications to documents.
Selecting and Activating Clinical Trial Sites
Identifying and onboarding suitable clinical trial sites is a critical step. Sites must have the patient population, infrastructure, and experienced personnel necessary to conduct the study effectively. Building strong relationships with investigators and their teams is vital for successful trial execution.
- Identify potential clinical trial sites based on patient population and research capabilities.
- Conduct site qualification visits to assess facilities, staff, and regulatory compliance.
- Negotiate clinical trial agreements and budgets with selected sites.
- Provide comprehensive training to site staff on the protocol, device use, and data collection.
Once IRB approvals are secured and sites are activated, the clinical trial can officially commence. This involves enrolling the first patient, initiating data collection, and rigorously monitoring the trial’s progress. Effective project management and close oversight are essential to ensure the trial adheres to the protocol, GCP, and all regulatory requirements. The launch of the clinical trial represents the culmination of a year of intense effort, marking a significant milestone in bringing a new MedTech innovation to patients.
By the end of month twelve, the MedTech startup will have successfully launched its clinical trial, with patients enrolled and data collection underway, moving closer to demonstrating the device’s clinical utility and impact.
| Key Milestone | Brief Description |
|---|---|
| Months 1-2: Concept & Design | Solidify core idea, market research, initial design, and IP strategy. |
| Months 3-4: Regulatory & Funding | Define FDA pathway, secure seed funding, and explore grant opportunities. |
| Months 7-8: Pre-clinical & QMS | Conduct pre-clinical testing and fully implement a robust Quality Management System. |
| Months 11-12: Launch Trial | Obtain IRB approval, activate sites, and officially launch the clinical trial. |
Frequently Asked Questions
The most critical step is solidifying the core concept, conducting thorough market research to define the unmet need, and initiating preliminary design while addressing intellectual property considerations. This foundational work ensures the device addresses a real problem and has market viability.
Early FDA consultation is highly important. It helps classify the device correctly, identify the appropriate regulatory pathway (e.g., 510(k), PMA), and understand specific data requirements. This proactive approach can significantly reduce delays and ensure compliance from the outset.
A robust QMS includes comprehensive documentation like Standard Operating Procedures (SOPs), work instructions, and quality records. It also involves internal audits, thorough personnel training, and preparation for external certifications like ISO 13485 to ensure adherence to quality standards.
MedTech startups should actively pursue seed funding rounds from angel investors and venture capitalists specializing in healthcare. Additionally, non-dilutive grants such as Small Business Innovation Research (SBIR) or Small Business Technology Transfer (STTR) grants are excellent options to explore.
A detailed clinical trial protocol is essential because it outlines the study’s objectives, design, participant criteria, data collection methods, and ethical considerations. It ensures scientific rigor, protects patient safety, and provides a clear roadmap for generating reliable and defensible data for regulatory review.
Conclusion
The journey From Concept to Clinical Trial: A 12-Month Roadmap for MedTech Startups in 2026 is undoubtedly challenging, yet highly achievable with meticulous planning and strategic execution. Each phase, from initial ideation and regulatory strategy to prototype development, pre-clinical testing, and ultimately clinical trial launch, builds upon the last, demanding precision and foresight. By adhering to this structured roadmap, MedTech startups can navigate the complex regulatory environment, secure vital funding, and develop safe, effective innovations that address critical unmet medical needs. The commitment to quality, compliance, and patient-centric development throughout this accelerated timeline is paramount for successful market entry and lasting impact in the evolving healthcare landscape.